MDR Documentation for Dental Technicians
MDR often means: Get the material, walk to the scanner, scan each batch individually. With DentaTool, this step is eliminated – you document correctly without interrupting your workflow.
Does this sound familiar?
DentaTool makes EU-MDR documentation so simple that it happens automatically.
Materials are entered centrally once. All batch numbers are available in the system and don't need to be scanned again each time.
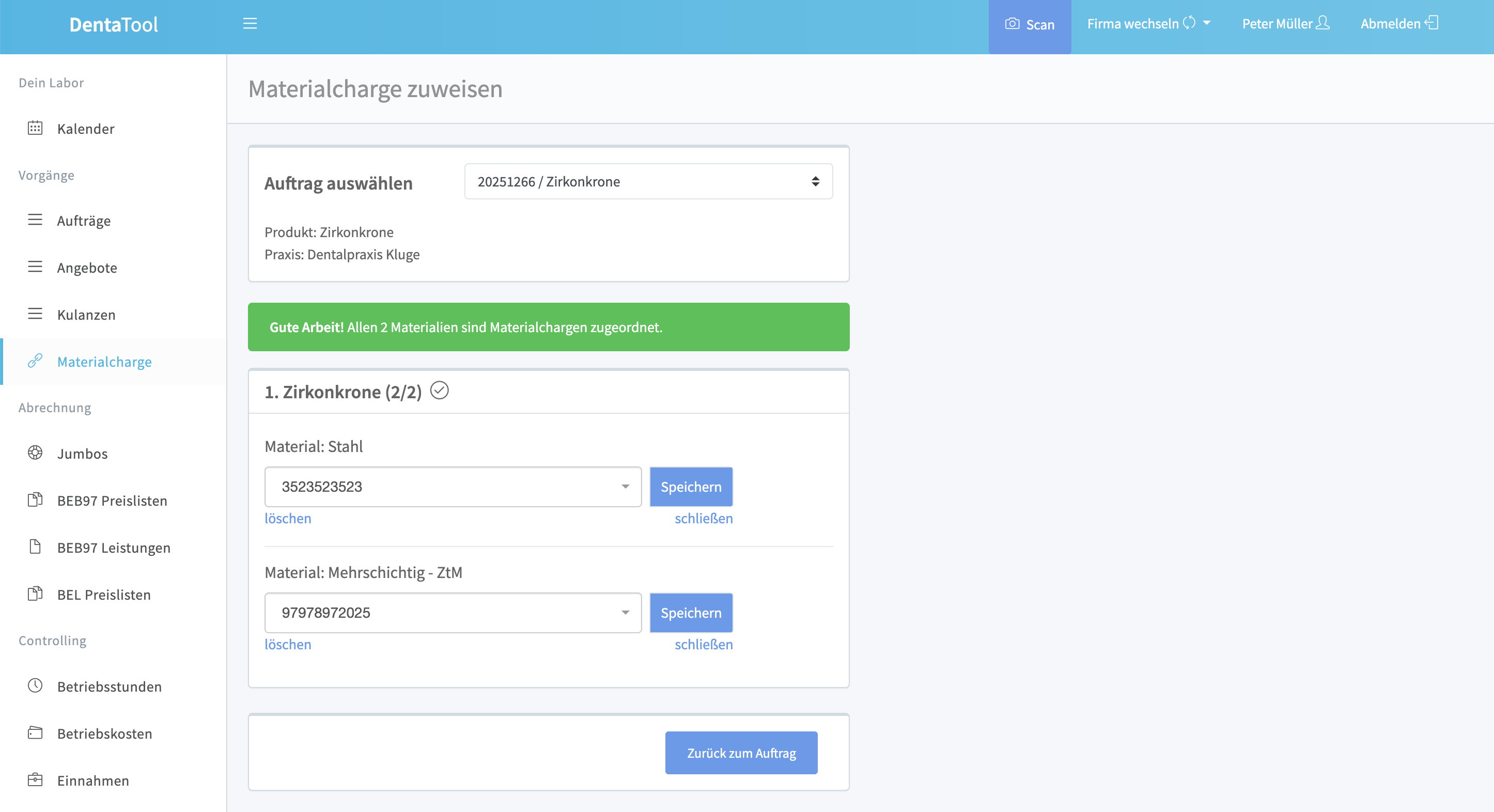
For standard orders, one click is enough to automatically assign open batches. No scanning, no searching, no interruption.
Manage open material batches centrally. See immediately which batches are still available and when restocking is needed.
Define for each product which materials are needed. During order entry, the system automatically knows what to assign.
Each batch is linked to each order. For inquiries or audits, you immediately find which material was used where.
Create declarations of conformity directly from DentaTool. All relevant data is automatically included.
See how easy batch tracking is with DentaTool.
| DentaTool | Manual Scanning | |
|---|---|---|
| Time per Order | One Click | Several Minutes |
| Workflow Interruption | None | Constant |
| Documentation Quality | Complete | Often Incomplete |
| Traceability | Automatically Linked | Tedious Search |
| Audit Preparation | Always Ready | Stressful |
See in a demo how DentaTool simplifies your MDR documentation.
No obligation · 30 minutes · no preparation needed
Find answers to common questions about this feature.